Replacing undesirable organic solvents with water-based reactions is a major preoccupation for chemists looking at more sustainable methods of active and intermediate synthesis. Dr Sarah Houlton examines some successes
Synthetic organic chemists tend to spend a lot of time ensuring there is no water in their reactions. It can interfere with many processes, perhaps by poisoning catalysts or reacting with reagents, so rigorous drying of glassware and keeping the atmosphere out is the order of the day. But in recent years, there has been a surge in interest in the exact opposite – carrying out reactions in water as a solvent.
The organic solvents in which most chemical processes are run are a big contributor to waste. Many have poor environmental credentials, particularly halogenated solvents such as dichloromethane. Although it can be possible to recycle solvents, reducing the quantities used and replacing them with more environmentally benign alternatives is a very important way in which processes can be made more sustainable.
It is sometimes possible to carry out reactions without any solvent at all if liquid reagents are involved, but these cases are in the minority. So, in practice, avoiding the worst offenders such as chlorinated solvents is the way ahead. However, if it is compatible with the process, water is the ultimate ‘green’ solvent and, in some cases, the reactions even proceed more rapidly and more selectively than their counterparts using organic solvents.
As a result, a good deal of effort is being put into developing reactions that run in water instead of organic solvents. The following are just a few recent examples of reactions that are important in industry where water-mediated variations have been investigated.
The industrial organic oxidation of alcohols to carbonyl groups still largely relies on chemistries that are less than sustainable, such as chromium-based reagents, explosive ones such as the Dess-Martin periodinane, or the sulfur-based Swern reaction. While the fundamental nature of these processes means they are often less water-sensitive, the routine use of cheaper reagents that have a lower environmental impact and a reduced requirement for organic solvents would be a significant advance.
As Roger Sheldon from the department of biotechnology at Delft University of Technology explains, the ultimate green oxidants are the oxygen in air and hydrogen peroxide, with water as the only by-product, in a metal-catalysed reaction.1 But oxygen and flammable organic solvents are not a good mix in terms of safety, and even using less than 10% of oxygen in nitrogen does not render the process completely safe on a large scale. Ideally, the reaction would be carried out in water, which would remove many of the safety concerns.
The reaction can be catalysed by palladium(II) salts, but the activity is low with the best turnover frequencies only about 10h–1, which is not good enough for an effective industrial process. The problem appeared to lie in the fact that the mechanism involves the reduction of the palladium to Pd(0) and then reoxidation by oxygen to Pd(II), but the Pd(0) had a tendency to aggregate, forming inactive palladium black. Ligands can be used to stabilise the Pd(0), and Sheldon’s group has investigated batho-phenanthroline disulfonate ligands, in combination with sodium acetate as a cocatalyst to help suppress the formation of palladium black. However, this system did not work in all cases, notably when the substrate alcohol also contains functional groups that include sulfur or nitrogen heteroatoms.
Kinetic studies led them to believe that the mechanism involved a hydroxyl-bridged dimer dissociating to the associated monomer, and this was the active catalyst. They thought that if they made more sterically crowded ligands derived from phenanthroline, these would dissociate more readily to the active complex, thus increasing its concentration and the reaction rate.
But, looking more closely at the reaction, they suspected that the very selective oxidation of the alcohol when an olefin was also present harked back to previous work by Moiseev,2 who had showed that palladium nanoparticles could selectively oxidise allylic C–H bonds in olefins.
So they decided to try pre-forming palladium nanoparticles, either by mixing palladium(II) trifluoroacetate and neocuproine in water and a cosolvent such as polyethylene glycol which both increased solubility and stabilised the nanoparticles, then reducing with hydrogen gas, or by adding a pre-formed complex of the two to hexanol dissolved in the water-cosolvent mixture. Palladium nanoparticles are formed in both cases.
The nanoparticles were then tested in a model oxidation reaction, the aerobic oxidation of 2-hexanol. The best conversions – more than 70% – were achieved with ethylene carbonate as cosolvent and 0.5 mol% of catalyst, with 8% oxygen in nitrogen in an autoclave. Further studies were carried out on more complex substrates, steroidal alcohols, and although the conversions were initially lower as the alcohols were less reactive, by increasing the catalyst loading to 5 mol% acceptable conversions were achieved in 4h (see Scheme 1).
Scientists at Tsinghua University in Beijing, China have developed a method for the synthesis of phenols, using a copper-catalysed oxidative hydroxylation reaction carried out in water.3 Typically nowadays, phenols are made from aryl halides, using palladium catalysts with phosphine ligands, but in some cases it is also possible to use the cheaper option of copper catalysts and non-phosphine ligands. The Chinese group has now found that arylboronic acids can routinely be oxidatively hydroxylated. The reaction takes place at room temperature in water, and the catalyst system is cheap – copper(II) sulfate plus 1,10-phenanthroline as ligand, and 3eq of potassium hydroxide to effect the hydroxylation.
Their first attempts using copper(I) oxide to oxidise 4-methoxy phenyl boronic acid in organic solvents were unsuccessful, with no reaction occurring, but by switching to a 1:1 mixture of water and DMSO they obtained about a 50% yield. In water alone, the yields were marginally better, and then when they added 1,10-phenanthroline, the yield rose sharply to 92%. They postulated that as copper(I) oxide cannot co-ordinate well with the ligand, the true active species was actually copper(II) which was being generated via a catalytic cycle, and so they tried the reaction using a variety of copper(I) and copper(II) species, stirring the reaction mixture at room temperature in the open air.
They determined that the optimum conditions involved 0.1eq CuSO4, 0.2 eq of 1,10-phenanthroline (which proved superior to all the other ligands they tried, such as TMEDA and L-proline) and 3eq of potassium hydroxide in water. A wide variety of different phenolboronic acids were investigated, with the lowest yield achieved 72%, and many were above 90%. And, unlike, the traditional halide chemistry, it works well for electron rich phenols. (Scheme 2).
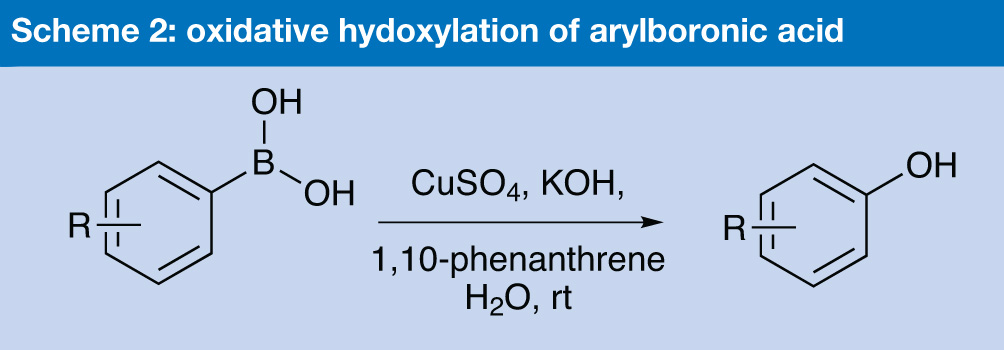
Scheme 2
Water has been used as the solvent in a synthesis of the analgesic fentanyl by Jonathan Williams and his group at the University of Bath, in collaboration with chemists from the University of Leeds.4 The key reaction is an iridium-catalysed alkylation of an amine by an alcohol, a general reaction they have developed that works well in water or ionic liquids, and converts primary amines into secondary ones, and secondary into tertiary.
Amines are usually alkylated by a substitution reaction using an alkyl halide, but overalkylation is a problem, and many alkyl halides have toxicity issues. However, the electrophilicity of simple alcohols is too low for the direct reaction to proceed easily. An alternative strategy would be temporarily to remove a hydrogen from the alcohol to give an intermediate aldehyde. This is electrophilic and reacts readily with the amine to give an imine, with the ‘borrowed’ hydrogen then being returned to it, giving the desired amine product.
The catalyst they used, [Cp*IrI2]2 or SCRAM, is typically used in toluene, but they found it worked well in both water and ionic liquids.5 The synthesis of fentanyl could be achieved in a single step, by reacting commercially available amidopiperidine with 2-phenylethanol, with 2 mol% of the catalyst and no added base. This was a higher catalyst loading than was necessary for unfunctionalised substrates, and they speculated that the amidopiperidine was co-ordinating to the catalysts as a bidentate ligand and making it less reactive (Scheme 3).
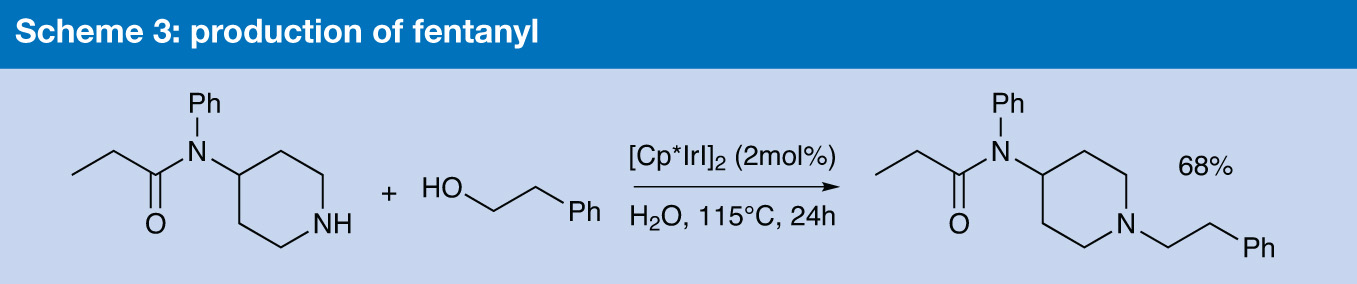
Scheme 3
Sometimes the organic solvent is necessary to provide solubility, and even if a reaction is compatible with water, it might also need some organic cosolvent to be added if reactants or reagents are simply too insoluble. If the problem is with the catalyst, then chemically modifying it to render it water soluble may be the answer.
The metathesis reaction is now a mainstay in the synthesis of pharmaceutical ingredients and intermediates, as it offers a powerful way of creating some challenging carbon skeletons under mild conditions. The ruthenium catalysts are generally tolerant of both air and water, and while it is possible to carry out these reactions in water, the catalysts’ low solubility usually has to be remedied by adding some organic solvent.
An alternative would be to incorporate a fragment of a surfactant molecule within the catalyst to improve its water miscibility, and a good deal of work has been carried out in this area by Bruce Lipshutz at the University of California, Santa Barbara.
Initially, they developed PQS, an amphiphile comprising polyethylene glycol, ubiquinol and sebacic acid components, that forms nanomicelles.6 It has a hydroxyl group that acts as a synthetic handle to attach it to other species that would otherwise be insoluble in water. An initial investigation to prove the principle, based on the first generation Grubbs–Hoyveda ruthenium catalyst, proved successful as it catalysed ring closing metathesis reactions to make disubstituted cycloalkenes. A derivative of the second generation catalyst would be more useful, as this would allow more complex metathesis reactions to be carried out, including cross metathesis.
As anticipated, this surfactant derivative of the second generation catalyst worked.7 It facilitated ring closing metathesis reactions in water at a loading of 2% by weight, at room temperature with a three-hour reaction time and with no exclusion of air from the reaction. Carbocycles and heterocycles containing nitrogen and oxygen both worked well.
Importantly, it was also easy to recycle the catalyst. All that was required was the addition of diethyl ether to the open reaction vessel to extract the product, brief stirring, and the organic layer is removed. This extraction was repeated once or twice and the solvent recovered, giving the desired product.
The catalyst remained in the aqueous phase, and could be re-used by adding further substrate. Even after eight recycles, they still obtained conversions in excess of 90%. The drift down in activity from 99% to 90% can be attributed to the normal behaviour of the ruthenium catalyst itself, and not to loss of catalyst during handling.
Cross metathesis reactions also proceeded well and under similar conditions, though the reactions take about four times as long. As the reaction takes longer, the residence time of the catalyst in the water increased, so the catalyst cannot be recycled as many times; adding further PQS after every three recycles almost returned the activity to its initial level.
Another surfactant-based strategy has been investigated by Grela’s group at the Polish Academy of Sciences in Warsaw in their work on metathesis reactions in water.8 They found that the emulsions formed by ruthenium complexes containing commercially available perfluorononanoic acid and a ruthenium carbonate complex in the presence of water and organic substrates were unstable and of poor quality. Their first alternative, using tocopherol as the surfactant, was similarly unsuccessful.
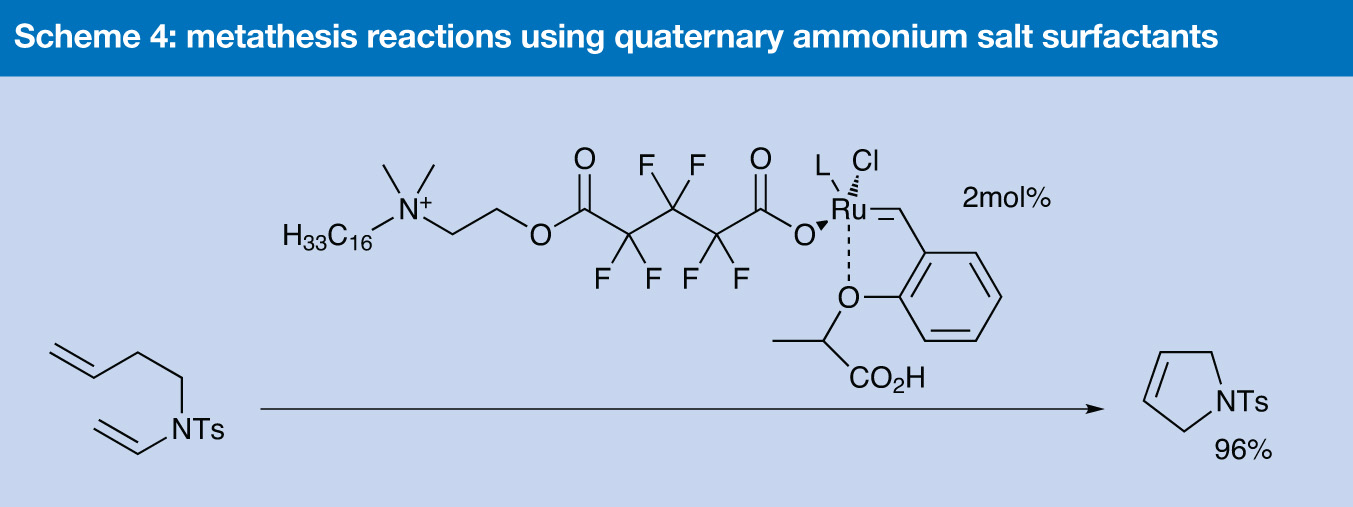
Scheme 4
However, better results were achieved when they turned to quaternary ammonium salt surfactants (Scheme 4). The resulting catalyst is insoluble in water, but forms a stable emulsion on stirring. It performed well as a catalyst in a variety of different ring closing metathesis and cross metathesis reactions in water as the only solvent. Importantly, the reactions proceeded well at room temperature in the open air, and yields were typically in excess of 90%.
Merck’s sitagliptin process wins EPA award
Even when it is not possible to replace organic solvents with a more environ-mentally benign alternative, it is still possible to make dramatic improvements in the sustainability of a process by altering the way it is carried out.
Scheme 5
Merck’s new biocatalytic process for the manufacture of the antidiabetic drug sitagliptin, developed with Codexis,9 was awarded the 2010 presidential green chemistry challenge award from the US Environmental Protection Agency for its improved efficiency and significant reduction in chemical waste by-products.
The previous synthesis involved an asymmetric enamine hydrogenation that was catalysed by a chiral rhodium complex, which needs to be carried out at high pressure. The stereoselectivity of the product could be better and it is contaminated by rhodium, which means that further separation and purification steps are required to increase both the purity and ee.
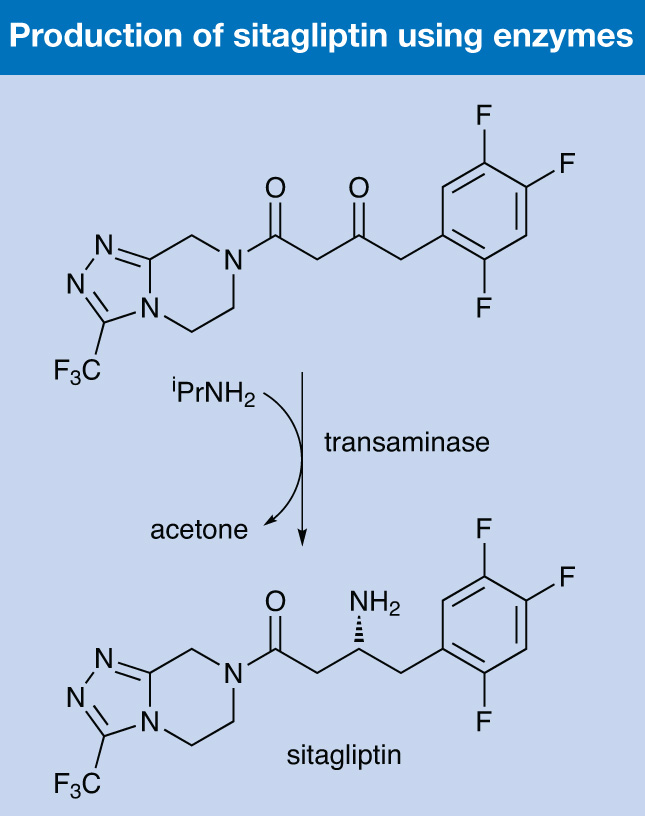
The chemists looked to a transaminase enzyme as a biocatalyst as an alternative. The starting R-selected transaminase worked on simple methyl ketones and small cyclic ketones, but did not hydrogenate the necessary substrate, so “substrate walking”, modelling and mutation was first used to find a starting point with at least some activity; this was then subjected to a directed mutagenesis programme to increase its activity to a level that was sufficient for an industrial synthesis.
The best engineered enzyme converted the prositagliptin ketone to sitagliptin at a concentration of 200g/l in an ee in excess of 99.95% – no amount of the wrong enantiomer was ever produced. The enzyme was used at 6g/l in 50% DMSO. It represented a 10–13% increase in overall yield over the original rhodium-catalysed process, a 53% increase in productivity in terms of kg/l a day, and a 19% reduction in total waste.
All heavy metals are eliminated, and no specialised high pressure hydrogenating equipment is needed, leading to an overall reduction in production costs.
references:
1. M. Mifsud et al. Tetrahedron 2010, 66, 1040
2. M.N. Vargaftik et al. J. Chem. Soc. Chem. Commun. 1985, 937
3. J. Xu et al. Org. Lett. 2010, 12, 1964
4. O. Saidi et al. Org. Proc. Res. Dev. 2010, 14, 1046
5. O. Saidi et al. Chem. Commun. 2010, 46, 1541
6. B.H. Lipshutz and S. Ghorai Org. Lett. 2009, 11, 705
7. B.H. Lipshutz and S. Ghorai, Tetrahedron 2010, 66, 1057
8. R. Gawin et al. Tetrahedron 2010, 66, 1051
9. C.K. Savile et al. Science 2010, 329, 305
