During the different stages of manufacturing and storages, the proteins can subject to chemically or enzymatically generated primary structure modifications, i.e. the post-translational modifications (PTMs), to contribute to the overall heterogeneity that forms part of the product quality attributes (PQAs) of the therapeutic proteins. The set of modifications that can affect drug’s efficacy and safety are classified as the critical product attributes (CPQs) and must be monitored in various quality control (QC) processes.
The Multi-Attribute Method (MAM) is an emerging application of using liquid chromatography (LC) coupled to high resolution accurate mass (HRAM) mass spectrometry (MS) for simultaneous monitoring of multiple PQAs. A major advantage of MAM is that it can detect the PTMs at the amino acid residue level, which is not possible with conventional profile-based methods using UV or fluorescence detection. The LC-HRAM-MS based MAM workflow involves initial enzymatic digestion of therapeutic protein into peptide of various length, followed by gradient separation using mobile phases made of water and acetonitrile with formic acid (FA) or trifluoracetic acid (TFA) as modifiers, and the HRAM MS analysis of the eluted peptides for determination of the sequence coverages that pinpoint the PTMs.
As an analytical methodology for QC in biopharmaceutical industry, the MAM must be robust and validated according to the strict regulatory requirements. The solvents and reagents used for mobile phases must be of high purity and void of contaminants that could cause interferences. In a most recent published study of MAM consortium interlaboratory study participated by more than 30 leading biopharmaceutical and LC-MS instrument vendor labs, the “chromatographic artifacts” and “contamination” were cited as two of the major reasons for result abnormality in peak detection. Figure 1 shows an example of two control (blank) runs using the mobile phase prepared from two different brands of LC-MS grade acetonitrile. As shown, the brand X LC-MS grade has an elevated background that could compromise the sensitivity. Furthermore, the repeating peak pattern arising from using the X brand acetonitrile as mobile phases could severely interfere with the peptide peak detection. The mass difference of 44 Da of these interfering peak pattern suggest they be the PEG or PEG-like contaminants. Thus, not all LC-MS grade solvents are made equal, and it is important to include the evaluation of solvents and reagents for mobile phases in implementing MAM methods.
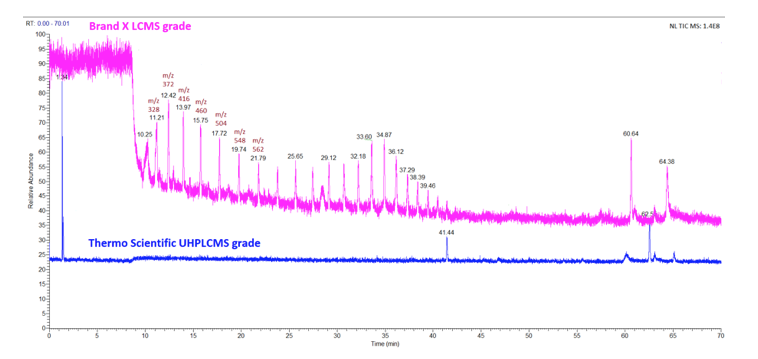
Figure 1. Control (blank) runs of an LC-MS MAM method using two brands of LC-MS grade acetonitrile for mobile phases: brand X (top in pink) and Thermo Scientific (bottom in blue). The mobile phase is a binary gradient of 0.1% formic acid in water and 0.1% formic acid in acetonitrile. The m/z of seven peaks showing PEG-like +44 Da are labeled (in brown).
Thermo Fisher Scientific offers two grades of solvents and blends that are fully tested for suitability for LC-MS analysis including LC HRAM MS MAM. The Fisher Chemical Optima grade LC-MS solvents and blends are made of high impurity, and precisely mixed with lot-to-lot consistency and tested with LC-MS. The Thermo Scientific UHPLC-MS grade solvents are a higher grade with more stringent specifications to meet the most sensitivity-demanding analyses. Both grades of solvents and blends have been fully tested to ensure low organic impurities including PEG like. In the newly introduced Thermo Fisher Scientific’s MAM 2.0 workflow, the UHPLCMS grade acetonitrile and water have been specified in preparing the mobile phase for the initial system performance evaluation test (SET).
References
1. Zhiqi Hao, et al, “Multi-attribute method performance profile for quality control of monoclonal antibody therapeutics”, J. Pharm. Biomed. Anal. 205 (2021) 114330
2. Trina Mouchahoir, et al, “New Peak Detection Performance Metrics from the MAM Consortium Interlaboratory Study”, J. Am. Soc. Mass Spectrom. 2021, 32, 913
3. Hao Yang, et al, “An integrated LC-MS system performance evaluation test for peptide mapping and monitoring”, Thermo Scientific Application Note 000336 (2021)
