It is becoming increasingly difficult to get new drugs to the market. While this is often attributed to pharma r&d organisations becoming less effective, the reality is far more complicated than that. For example, regulators are demanding an ever increasing number – and size – of clinical trials to prove that drugs are both safe and efficacious, which makes bringing a drug to patients far slower and more expensive.
And the ‘easy’ targets have already been hit. Heart disease, for example, is now so well served by drugs such as ACE inhibitors, calcium channel blockers, statins and diuretics – all of which are available as very cheap generics – that it is difficult for new drugs to compete. Not only would cost be an extremely big disincentive to prescribing them, regulators now demand not just proof that the drugs work and are safe, but proof also that they are better than any existing alternatives. Gone are the days when the annual list of approved drugs would be 50-strong, and would include half a dozen different molecules all directed at the same target; the “me-too” is becoming a thing of the past.
That said, there are numerous diseases where there is still a great deal of therapeutic need. While many of these are niche orphan diseases where patient numbers are limited, there are some widespread conditions that are not adequately treated or controlled by existing medicines. One of these is Type II diabetes. Its incidence is increasing, thanks to the modern indolent Western lifestyle with associated bad diet, yet there is still a need for improved drug treatments. This is not least because some of the most effective drugs currently available to patients – the glitazone drugs Avandia (rosiglitazone, GSK) and Actos (pioglitazone, Takeda) – face ongoing concerns about their cardiac safety.
Type II diabetes is one of the few common conditions for which the European Medicines Agency gave a positive opinion on new drugs last year – not just one, but two, which act at different targets. The first of these, liraglutide (Victoza, Novo Nordisk), is a long-acting analogue of glucagon-like peptide-1, or GLP-1, a biologic that is given as a once-a-day subcutaneous injection. GLP-1 itself has a half-life of only about an hour, and thus is not itself therapeutically useful. Liraglutide is a palmitic acid derivative of the natural peptide, which activates the GLP-1 receptor. This ultimately increases the release of insulin in the presence of elevated levels of glucose, and decreases glucagon secretion in a glucose dependent manner.
The second, saxagliptin (Onglyza, Bristol-Myers Squibb and AstraZeneca), is actually the third gliptin drug to gain a positive opinion from the regulators, after Novartis’ vildagliptin (Galvus) and Merck’s sitagliptin (Januvia), although the former was knocked back by the US FDA. These drugs are dipeptidyl peptidase-4 inhibitors, which work on the same cascade as liraglutide, by improving the secretion of insulin from the b-cells from the pancreas in response to an increase in blood sugar, and also decrease the output of glucagon from the a-cells. This is caused by a decrease in the breakdown of GLP-1, so it reaches a more normal level, and the alteration in glucagon levels correlates linearly with an improvement in glucose tolerance.
One heart condition that is poorly served by drug treatment is atrial fibrillation, and Sanofi-aventis’ dronedarone (Multaq) was designed to address this problem; the main alternative, amiodarone (Cordarone, Wyeth) is an old drug that is dogged by side-effect issues. The heartbeat is controlled by an electrical impulse that starts in the right atrium, but in a patient with atrial fibrillation this impulse sometimes goes awry and the heart goes out of rhythm. The drug’s precise mechanism of action is unknown, but it blocks sodium, potassium and calcium channels, as well as having antiadrenergic effects, and in clinical trials was shown to increase significantly the length of time between attacks. However, in January an initial decision from NICE indicated that it would not be recommended for reimbursement in the UK, despite the absence of a well tolerated alternative.
A huge amount of discovery effort is being put into finding new treatments for cancers; this is another area where NICE’s pronouncements have a significant impact, as frequently they knock back approved cancer drugs as they are so expensive. However, research continues unabated, and several anticancer agents were given the go-ahead by EMA last year.
Vinflunine (Javlor, Pierre Fabre) is a fluorinated vinca alkaloid that was developed to treat bladder cancer, and was the first drug to get a positive opinion in Europe for this disease. It is indicated as monotherapy to treat advanced or metastatic transitional cell carcinoma of the urothelial tract after failure of platinum-based therapy. It is a microtubule inhibitor, like the taxanes.
Everolimus (Afinitor, Novartis) is a rapamycin derivative, and – like rapamycin itself – acts as an mTOR, or mammalian target of rapamycin, inhibitor. It was given orphan drug status by EMA for kidney cancer, and has been approved for the treatment of patients with advanced renal cell carcinoma whose disease has progressed despite treatment with vascular endothelial growth factor targeted therapy.
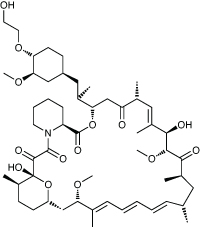
Everolimus
AstraZeneca’s gefitinib (Iressa) has been the subject of much controversy over its efficacy, but finally got the go-ahead in Europe last year for the treatment of locally advanced or metastatic non-small cell lung cancer in patients who have previously received chemotherapy. It is a selective inhibitor of epidermal growth factor receptor’s tyrosine kinase domain. EGFR is overexpressed in several human tumours, notably lung and breast cancers, and a mutation in this domain is believed to be responsible for activating anti-apoptotic mechanisms. It binds to the ATP binding site of the enzyme, inhibiting its effect in activating the Ras signal transduction cascade.
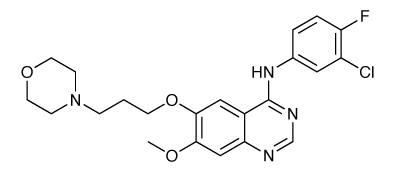
Gefitinib
Malignant ascites can occur in patients with metastatic cancer, and is an accumulation of fluid in the peritoneal cavity. A new treatment, catumaxomab (Removab, Fresenius) works by binding to the CD3 and EpCAM antigens; it consists of one half each of antibodies against the two antigens, which also binds to an Fc receptor on a cancer-killing cell. EpCAM, or epithelial cell adhesion molecules, are present on the surfaces of a variety of different cancer cells. An immunological reaction against the tumour is triggered by binding to a cell bearing EpCAM by one arm, a T-lymphocyte by the other arm, and an antigen presenting cell such as a macrophage or an NK cell by the heavy chains of the combined antibodies. This can remove cancer cells from the peritoneal cavity, and thus reduces the ascites that are produced in response to the cancer burden.
Therapeutic options to treat autoimmune conditions such as rheumatoid arthritis are growing, thanks to the introduction of a variety of monoclonal antibody drugs in recent years. Two more were given the thumbs up by EMA in 2009: certolizumab pegol (Cimzia) from UCB, and Contocor’s golimumab (Simponi), both of which target tumour necrosis factor a. Certolizumab pegol is a pegylated FAB' fragment of a humanised TNF inhibitor antibody, and has been approved for the treatment of rheumatoid arthritis. Golimumab is a human IgG1k monoclonal antibody that is specific for TNF-a, and is indicated for the treatment of three autoimmune diseases – rheumatoid arthritis, psoriatic arthritis and ankylosing spondylitis.
Bone marrow transplantation is a cornerstone of the treatment of blood cancers such of leukaemia. Healthy haematopoeietic stem cells are introduced after the native bone marrow is killed off. The stem cells have to be mobilised before transplantation, and plerixafor (Mozobil, Genzyme) is a new agent with this activity. It acts at the CXCR-4 receptor, by inhibiting the interaction of this receptor and stromal cell-derived factor 1.
Plerixafor was originally designed as a drug to treat HIV, and another drug initially developed for another indication, thiotepa, has also been given a positive opinion in HSC transplants. Marketed by Adienne as Tepadina, it is designed to be used as a conditioning treatment for the cells prior to transplantation, and was initially used to treat breast, bladder and ovarian cancers.
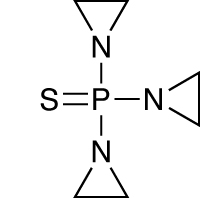
Thiotepa
Benign prostatic hyperplasia, or BPH, is a very common condition in men as they age and causes problems with urination. Silodosin (Urorec, Recordati) is a selective inhibitor of the a-1 adrenergic receptor, and by blocking these receptors in the bladder neck and prostate – as well as blocking the neurotransmitter norepinephrine from binding to its receptor – it relaxes the smooth muscle tissue in both prostate and bladder, thus improving urine flow and reducing the symptoms of BPH.
Meanwhile, a new treatment for constipation, Movetis’ prucalopride (Resolor) is indicated for the symptomatic treatment of chronic constipation for women in whom laxatives are insufficiently effective. It is a 5-HT4 agonist with better selectivity than older agents, and accelerates gastric emptying.
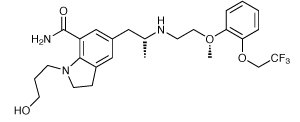
Silodosin
The growing trend in the western world for women to have children later in life has led to an increase in demand for fertility treatment to offset the natural decline in fertility that takes place as they age. Assisted reproduction techniques, such as in vitro fertilisation, rely on the harvesting of eggs, either from the woman herself or a donor, and before this is done a hormone treatment is given to accelerate the development of follicles into eggs and increase the number that can be collected. Merck’s corifollitropin alfa (Elonva, developed by Merck legacy company Organon) is given in combination with a gonadotropin releasing hormone antagonist to stimulate the ovaries. This first-in-class drug is a sustained follicle stimulant, which can initiate and sustain multiple follicular growth with a single subcutaneous injection.
Standard treatment is with daily injections of recombinant follicle stimulating hormone preparations, and corifollitropin is designed to replace the first seven days of these injections.
At the other end of the fertility scale – emergency contraception – HRA Pharma’s ulipristal acetate (Ellaone) provides a new alternative. It is a selective progesterone receptor modulator, and extends the window for effective contraception from three to five days after unprotected sex.
Its efficacy is comparable to levonorgestrel, at about 60%. The drug is both an antagonist and a partial agonist for the progesterone receptor, binds to the glucocorticoid receptor, and is also thought to delay the maturation of the endometrium and delay, or even block, ovulation.
Osteoporosis is very common in postmenopausal women, and two new treatments were approved. Amgen’s denosumab (Prolia) is a fully human monoclonal antibody that binds to RANKL, or receptor activator of nuclear factor k B ligand; RANK itself causes preosteoclasts to be converted into mature osteoclasts. This binding inhibits the maturation of osteoclasts, which thus protects the bone from degrading. It is given every six months by subcutaneous injection. The second osteoporosis drug to be given the green light last year is Wyeth’s bazedoxifene (Conbriza). Unlike denusomab, it is a small molecule, and is a selective oestrogen receptor modulator.
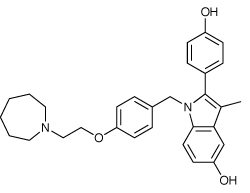
Bazedoxifene
Patients with chronic kidney failure are at risk of developing hyperphosphataemia, and sevelamer (Renvela) from Genzyme prevents dietary phosphate from being absorbed by the gut by binding to it. Conversely, patients with conditions such as congestive heart failure commonly suffer from hyponatraemia, or low sodium levels, as a result of diuretic treatment.
Otsuka’s tolvaptan (Samsca) is a competitive arginine vasopressin receptor-2 antagonist, and when given alongside existing diuretics it increases the excretion of excess fluids while also improving blood sodium levels.
Capsaicin is an active compound found in chilli peppers that causes a burning sensation on the skin. It is an agonist of the transient receptor potential vanilloid 1 receptor, or TRPV-1, an ion channel receptor complex that is expressed on nociceptive nerve fibres in the skin. Qutenza from NeurogesX is a patch formulation of capsaicin that is now indicated for the treatment of peripheral neuropathic pain in non-diabetic adults.
Eslicarbazepine acetate (Exalief, BIAL-Portela) is a new treatment for epilepsy. It is a prodrug that is activated to eslicarbazepine in the body – a metabolite of the existing epilepsy drug oxcarbazepine. Unsurprisingly, it has the same mechanism of action as the parent drug – it blocks voltage gated sodium channels, and is indicated as an adjunctive therapy in adults with partial onset seizures.
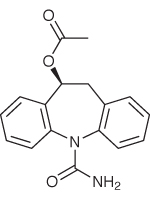
Eslicarbazepine
orphan drugs
Several new drugs to treat orphan diseases were also approved – two of them to treat the same condition. Canakinumab (Ilaris, Novartis) and rilonacept (Arcalyst, Regeneron) are both indicated for the treatment of cryopyrin-associated periodic syndromes. These are a group of rare genetic syndromes that are usually caused by mutations in the NLRP-3 gene.
Canakinumab is a human monoclonal IL-1b antibody that neutralises IL-1b’s activity and blocks its interaction with IL-1 receptors. Rilonacept is a dimeric fusion protein consisting of the ligand binding domains of the extracellular portions of the human inter-leukin-1 receptor component and IL-1 receptor accessory protein, linked to the Fc portion of human IgG1, and acts as an interleukin-1 inhibitor.
A far simpler molecule, amifampridine – 3,4-diamino-pyridine – has been approved as EUSA Pharma’s Zenas to treat Lambert-Eaton myasthaenic syndrome. In this autoimmune disorder, voltage gated calcium channels on the presynaptic membrane at the neuromuscular junction are inhibited, stopping the release of acetylcholine and, ultimately, preventing muscle contraction. It is often associated with non-small cell lung cancer. Amifampridine blocks potassium channel efflux, increasing action potential duration, thus leaving calcium channels open for longer.
Another orphan product, eltrombopag (Revolade, Glaxo-SmithKline), is designed to treat chronic immune thrombo-cytopoenic purpura. The small molecule drug is an agonist of TpoR, the receptor at which the hormone thrombopoeitin acts, and causes an increase in platelet counts.
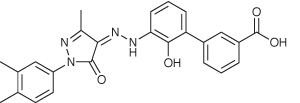
Eltrombopag
Finally, TiGenix’s Chondro-Celect is the first advanced therapy medicinal product to be given a positive opinion by EMA. This is a cell-based medicine that consists of cartilage forming cells, chondrocytes, that have been expanded in vitro, and which is designed to repair cartilage defects at the end of the thigh bone in the knee. Cartilage damage is common, but its ability to autoregenerate is limited. The cells are taken from a small biopsy of the patient’s own healthy cartilage, grown outside the body, and then reimplanted during cartilage surgery.
EMA’s new Committee for Advanced Therapies, a committee that was formed to reflect the growing interest in these types of treatment, evaluated the treatment. It seems inevitable that more advanced therapies like this will reach the market in coming years, providing treatments for patients whose current options are limited or ineffective.
