What is mRNA and its importance within the vaccine field?
mRNA is part of the DNA (deoxyribonucleic acid) family. mRNA, or messenger Ribonucleic Acid, plays an integral role in protein synthesis. In addition, its counterpart DNA, is a double stranded molecule, whereas mRNA, is single stranded.
mRNA vaccinology is advantageous because it offers adaptability in both the number and type of antigenic determinants it encodes. Its versatility means that mRNA can be described as an attractive source of antigen, and the nature of mRNA leads to different advantageous benefits.
Lyo: The right equipment to ensure the best outcome
As previously mentioned, the advantageous nature of mRNAs has facilitated their usage within the vaccine production industry. To achieve the goals set during the development phase, it is imperative all equipment used for R&D ensures a sufficient level of data is captured, allowing process scientists to create a defined ‘design space’ for the vaccine. Equally, the process must be scalable, so any piece of information retrieved at the R&D stage should also be available at the commercial level – enabling operators to conduct full scale-up / down process reviews, when required.
A data rich environment, (PAT benefits) allows for continuous process monitoring and improvement as expected by regulatory agencies, with focus on allowing lyo scientists to better understand different facets of the freeze drying process. The ability to identify that key metrics within a process do not exceed a pre-designated threshold for example, are paramount in contemporary manufacturing environments.
The above allows companies the opportunity to thoroughly investigate batches where possible unforeseen deviations may have arisen, but not to the extent where they move the sample outside their defined ‘design space’.
A Qualified Person, QP, on-site is then able to review and confirm product data remained within its safety margin and is therefore released, rather than rejected. From a company perspective, by purchasing equipment capable of supplying such a volume of key data, may be the difference between being able and confident to release a batch that could be worth >£1M, or rejecting it.
In a similar fashion, selecting a freeze dryer with the capability to stringently control the nucleation point of the material via ControLyo can improve inter and intra-batch product quality through a QbD (quality by design) approach. This aids in facilitating benefits to both the product/ sample such as product in all vials having the same nucleation temperature and timestamp during the freezing phase, including those with a thermocouple; this in turn reduces stress on biologicals, allows larger ice crystal sizes, minimises dry layer resistance, and offers a more uniform, shorter reconstitution time.
From an operational and finance position, the benefits are vast. By way of illustration, the removal associated with the risk of foreign material entering vials, enhances quality which leads to lower rejection rates. Increased production capacity through shorter cycles (therefore a reduction in costs), in conjunction with other benefits like improved safety of the cycle and scalable technology, from R&D up to commercial scale equipment used for manufacturing.
Aseptic Fill/Finish: The right equipment to ensure the best outcome
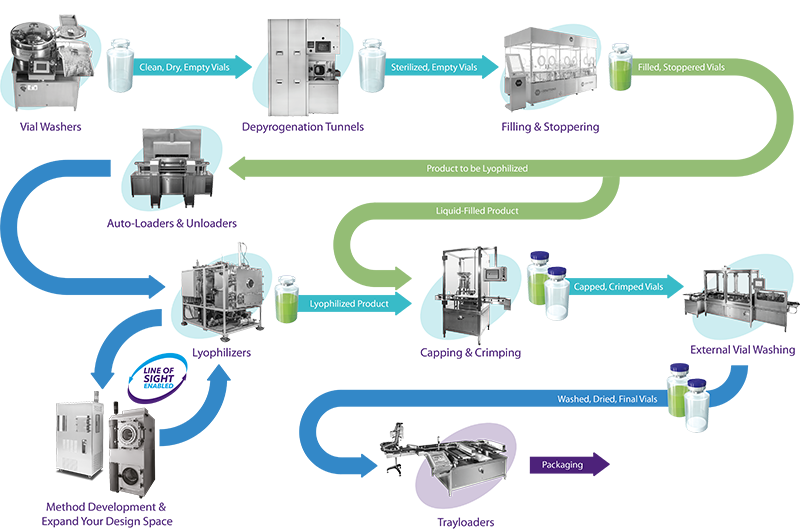
When approaching the fill/finish aspect of the manufacturing cycle, especially for vaccines, it is important to understand the stringency of filling tolerance required. Other areas of paramount importance being the removal of possible liquid ‘splashing’ during the container fill stage. Intuitive feedback, such as no container, no fill, check weighing, and integration within isolator conditions are vital, along with the need for strict software data integrity so the equipment and process is 21 CFR Part 11 compliant.
Even so, it remains highly desired that fill/finish equipment offers a good level of flexibility. Vaccine production may demand a particular vaccine within a 2R vial, the next in a 10R vial, the next within a ‘ready-to-use’ (RTU) syringe, cartridge, or vial.
Machine versatility and ease of use is therefore crucial, as the biopharmaceutical industry transitions to smaller and more targeted aseptic batches for personalised patient treatments. In doing so, containers such as vials, syringes, cartridges of differing sizes are relatively common, with RTU containers becoming increasingly favoured within industry.
In addition, aseptic filling must accommodate a nest with empty containers placed within an ISO standard tub (ensuring safe transportation), de-bagged automatically, pre-heat the lid during the de- lidding process, allowing the containers to be sorted within the RABS environment and readied for filling.
In conclusion, due to the nature of mRNA vaccine production, whether looking at the lyo/ freeze drying or liquid fill and fill elements of vaccine processing, equipment flexibility and data availability/ integrity is essential.
Selecting the correct machinery for the desired needs helps ensure batch rejection rates are as low as possible, whilst allowing multi-faceted vaccine formats to be manufactured on the same system (albeit via implementation of change parts), to maximise efficiency from both an operational cost and space / unit footprint, perspective.
To discover more about how aseptic fill-finish processing lines can support the production of mRNA vaccines or to discuss your requirements, contact one of our specialists today.

