Its rich heritage of financial security, strong privacy laws and a pragmatic regulatory approach have made Switzerland a vibrant hub for the blockchain and distributed ledger technology (DLT) behind crypto assets. The country both fosters innovation and offers a reliable regulatory framework.
Initiated in the city of Zug, “Crypto Valley” has established a worldwide reputation as a catalyst for new blockchain business ventures, with early movers such as Ethereum and Bitcoin Suisse paving the way.
Meanwhile, Crypto Valley has spread far beyond the borders of Zug, now comprising leading blockchain ecosystems in Zurich, Geneva, Neuchâtel, Ticino, Basel, Vaud, Lucerne and Bern.
In 2021, the new DLT Framework came into force, giving Switzerland one of the world’s most forward-looking legislation by creating space for innovative projects and entirely new business models in the digital and decentralised world.
Sirpa Tsimal, Director of Investment Promotion at Switzerland Global Enterprise, comments in the 2022 Swiss Biotech Report: “With the rise of new technologies such as blockchain and artificial intelligence (AI), the pragmatic regulatory approach of the Swiss authorities has become an important asset."
"Swiss laws, unlike those in many other countries, are formulated in a technology neutral way, which means that companies implementing novel business models and emerging technologies can operate within existing law and enjoy legal certainty. This progressive regulatory environment laid the foundation for the now famous Swiss Crypto Valley.”
It all started with a few pioneering crypto entrepreneurs who were looking for a jurisdiction that would allow them to experiment with a new technology: blockchain. Their success attracted like-minded companies and, soon, Switzerland had established a worldwide reputation as a catalyst for new blockchain business ventures with a variety of new applications that go far beyond the finance sector.

In the life science industry, Switzerland’s longstanding expertise has produced a strong ecosystem across the entire value chain that has always been attractive to biotech companies.
Sources of innovation in this sector increasingly stem from other business models, such as the application of processes, databases and data analytics that allow the smart application of therapies or selection of patient groups. This allows more effective and targeted therapies for personalised medicine and more focused drug development.
Introducing PharmaLedger
Capitalising on this environment of innovation, the Basel-based pharmaceutical multinational Novartis proposed the PharmaLedger project to explore potential applications of blockchain technology in the medical industry. The first application is set to provide patients with electronic medical leaflets.
“PharmaLedger was initiated under the EU’s Horizon Europe (Innovative Medicines Initiative) programme and included 30 member organisations,” says Daniel Fritz, Executive Director of the PharmaLedger Association: “It comprised 12 global pharmaceutical companies and 18 public and private entities, including technical, legal, regulatory, academia, research and patient representative organisations.”
“The goal of the project was to provide a widely trusted platform that supports the design and adoption of blockchain-enabled healthcare solutions while accelerating the delivery of innovation that benefits the entire ecosystem — from manufacturers to patients.”
"Another goal was to ensure the sustainability of the project outcomes, which resulted in the creation of the PharmaLedger Association, a Swiss Association registered in Basel.”
Marco Cuomo, Manager, Applied Technology Innovation at Novartis, adds: “The idea behind PharmaLedger was sparked in late 2017 during a conference in Switzerland. Crypto Valley, with all its innovative and creative start-ups, the various universities with blockchain centres of excellence and the strong community around blockchain topics in Switzerland is a key element to create and foster ideas and bring them to life."
"Not only at the beginning of our journey with PharmaLedger, but especially during the ups and downs of such a huge undertaking, it was and still is extremely helpful to tap into such a large and diverse pool of experts to discuss and solve problems.”

Describing PharmaLedger as a digital trust ecosystem (DTE) for healthcare, “it’s created and governed by those who work with it and within it,” says Dan.
“The blockchain network in Switzerland and across the world is very inspiring and has allowed us to think about new ways of governance between classic and completely decentralised approaches. We can learn from others, but we can also give back a lot — particularly in terms of sustainability and exploiting future potential — which is what makes it so exciting.”
Delving deeper
During a recent conference call with Dan and Marco, I ask about PharmaLedger’s key objectives. “There were many aspects to consider,” notes Marco, “such as data siloes and the lack of provenance, visibility and transparency, as well as inefficiencies (and waste) throughout the supply chain. Furthermore, the risk of cybercrime is increasing, counterfeit medicines are rife and there’s still work to be done regarding patient centricity: identities are often monetised.”
An opportunity clearly existed for an open innovation platform such as a DTE. Automation and disintermediation could be used to increase the efficiencies of healthcare systems, patients could be better protected — and empowered — by detecting falsified medicines and the secure sharing of data could accelerate innovation and create value.
“A collaborative mindset was vital, though,” says Dan: “It’s probably a more important element than the concept of blockchain itself.”
“The PharmaLedger Association enables and accelerates digital transformation and value creation for patients and stakeholders with a common platform for collaborative innovation and trusted transactions in the healthcare DTE.
The preferred way to connect, protect, empower, build and sustain the modern digital healthcare experience. It aims to be the preferred way to connect, protect, empower, build and sustain the modern digital healthcare experience.”
Within a framework that ranges from patients and service/healthcare provides to manufacturers, regulators and academia, Dan feels that use cases can become valuable solutions to healthcare problems. ‘We’re looking to build to connect as opposed to build to replace,” he says.
Membership benefits
When asked about the advantages of being part of PharmaLedger, Dan is keen to highlight the possibilities of accelerated digital innovation and learning in your organisation, being able to “add your voice and expertise” to digital healthcare development and pool resources and risk with like-minded partners to realise increased benefits.
There are gains to be had from collaborating with small and large organisations, as well as promote your existing solution within the association’s network and sourcing healthcare partners to work on novel projects.
PharmaLedger’s research will be divided into Core and Sponsored projects. The former are funded directly by the association’s “xLab.”
This programme provides seed funding for high risk, high-reward collaborative proposals in the area of innovative technology; for the latter, members and non-members can sponsor more in-depth collaborations between xLab participants and PharmaLedger Association member companies on specific mutual research interests and use cases.
xLab works closely with both the pharmaceutical organisation and the xLab participants to understand what the interests are and cultivate the collaborations by consistently facilitating the development process.
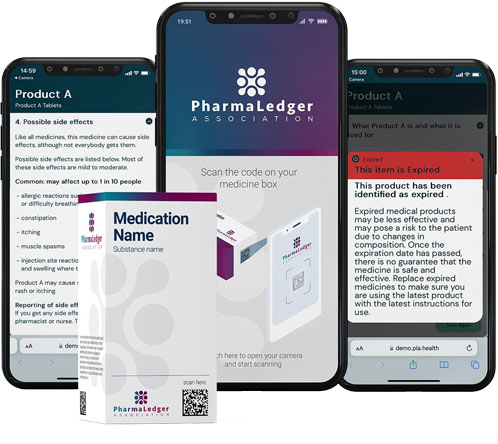
A case in point is the ePI (electronic product information) app. Key focus points are providing easier to read information in structured and searchable text. Patients just scan a barcode on their medication pack (with a smartphone) to get a digital leaflet.
The technology is system agnostic, regardless of the manufacturer, and allows users to access the most-up-to-date information (expiration date, recall notifications, etc.) from a trusted and secure source. Future versions will include video content, adherence reminders, adverse event reporting and links to other healthcare apps.
“In this instance, blockchain simply serves as the common ‘resolver’ for the patient to access the latest product information,” says Dan: “Not only does the app enhance security by eliminating the use of a central server, it also ensures that there is no single point of failure and promotes availability."
"Naturally, patient data privacy is maintained, as is the integrity of the product information.” He adds: “Furthermore, the app is scalable and flexible, opening the door to value-adding digital services in the future.”
Looking ahead
There’s work to do, concedes Dan, but notes: “We’re aware of the hurdles that need to be overcome. Between the drug maker, wholesaler and the pharmacy, there’s a lack of interoperability and an inconsistent availability of downstream ecosystem data. Information is siloed and there are too many IT interfaces, leading to inconsistent demand management and inaccurate forecasting.”
“The blockchain-enabled future is one of 100% finished goods traceability,” he says, concluding: “PharmaLedger puts patient safety at the heart of everything we do by enabling improved collaboration among ecosystem partners to support secured, trusted data sharing and by delivering end-to-end visibility of downstream data to optimise supply chain operations.”
Discover more about the PharmaLedger Association during their presentation at Manufacturing Chemist LIVE!
